 
Activators increase the rate of electrophilic aromatic substitution and deactivators decrease it: The ones that withdraw the electron density are deactivators. /Benzene_Toluene_and_ortho-meta-and_para-xylene.svg-58a39a425f9b58819c613292.png)
In short, the groups that donate electron density to the ring and make it electron-rich are activators. Halogens deactivate the aromatic ring, yet they direct the electrophile to the ortho and para positions.īut why? How does that happen, right? Well, first a few words on the activators and deactivators. 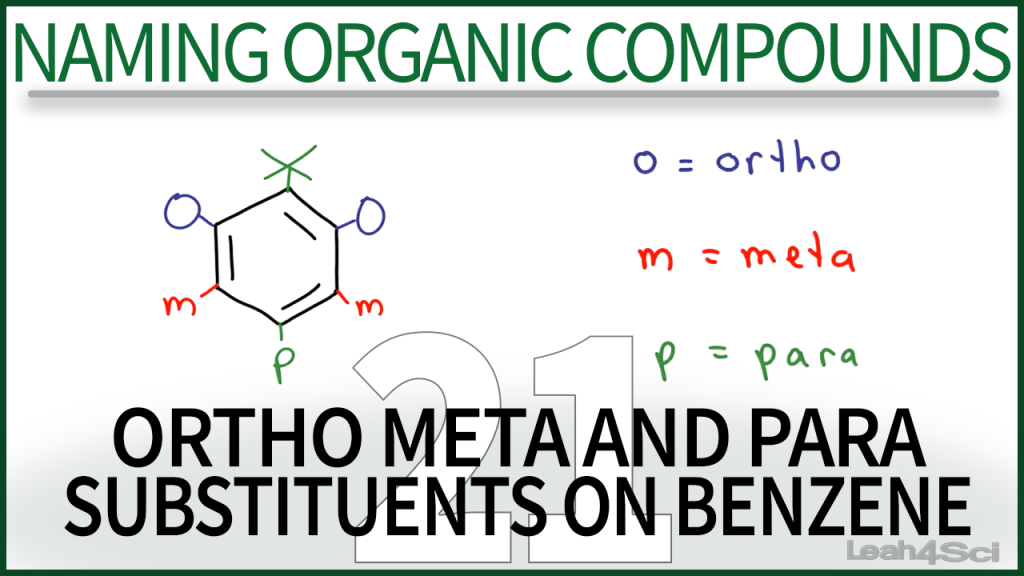
To which one the group belongs, depends on how it stabilizes or destabilizes the transition state of the electrophilic substitution reaction.Īctivating groups (the ones that stabilize the transition state and make the reaction faster) turn out to direct the electrophile to the ortho, para positions, while the deactivating groups put them in the meta position, except for the halogens! There are three relative positions for a disubstituted benzene ring: ortho, meta, and para.Īll the functional groups are divided into ortho-, paraor meta-directors. When a benzene ring with a substituent undergoes an electrophilic aromatic substitution, the electrophile is installed on a specific position(s) depending on the substituent. What are Ortho-, meta and para directors? Now, let’s discuss this a bit more in detail by first doing a quick overview of the ortho, para and meta directors first. Here is short summary answer as to why halogens, being deactivators, are still ortho, para directors: Halogens are ortho, para directors, however they are also deactivators! 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
